Summer 2021 (Volume 31, Number 2)
Top 10 Things You Should Know About the Eye in Your Rheumatology Practice
By Vanessa Ocampo, MD, FRCPC
Download PDF
Rheumatology is a subspecialty that doesn't just treat
diseases that affect the musculoskeletal (MSK) system.
One of the most common extra-musculoskeletal
(EMSK) anatomical sites involved is the eye. The following
are things we should know about the intersection of these
two subspecialties, that you may find helpful.
-
Knowledge about basic ocular anatomy and the structures that can be affected by inflammatory conditions will help us to recognize them more easily, perform an appropriate evaluation, and prompt referral to the ophthalmology team. (see Figure 1).1
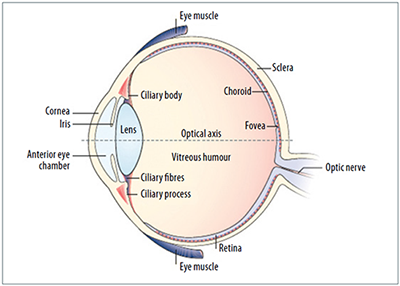
Figure 1: Overview of the Eye
The outer layer of the eye is composed of the cornea and the sclera. The inner layer consists of the retina. In conditions such as uveitis, dividing the eye anatomically by segments can help to classify the disease more easily (see Figure 2):
- Anterior: iris
- Intermediate: ciliary body, anterior vitreous, pars plana
- Posterior: choroid, retina and optic nerve
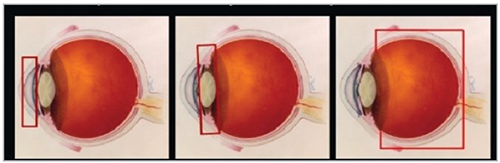
Figure 2: Classification of Uveitis
-
The most common ocular manifestations of rheumatic diseases include keratoconjunctivitis sicca, anterior uveitis (AU) and scleritis among others.2,3
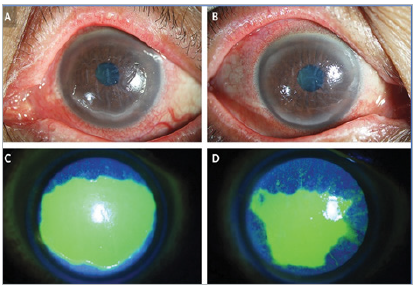
Figure 3:
Keratoconjunctivitis
Sicca in Sjogren’s4
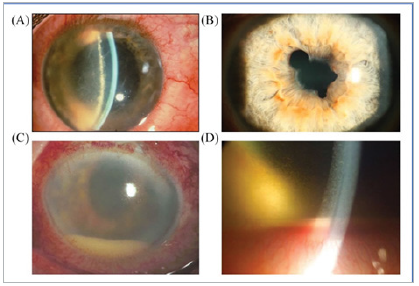
Figure 4:
Acute Anterior Uveitis
(AAU): A) Ciliary
injection; B) Synechia;
C) Hypopyon; D) Fibrin
coating at the front of
the lens5
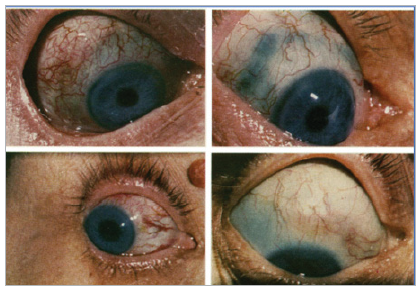
Figure 5: Scleritis in
Rheumatoid Arthritis6
-
As rheumatologists we should recall that there are multiple eye manifestations of rheumatologic conditions such as the ones listed in Table 1. 7,3,8,9,2
Table 1: Ophthalmologic and Rheumatologic Associations
| Ophthalmologic diagnosis |
Rheumatologic association |
| Orbital inflammatory disease |
GPA*, sarcoidosis, IgG4-related disease, Sjogren's syndrome, IBD*,
Behcet’s disease, RA*, adult-onset still’s disease, amyloidosis,
histiocytic disorders |
| Keratoconjunctivitis sicca |
RA*, SLE*, scleroderma, Sjogren’s syndrome, graft vs host disease, RPC* |
| Episcleritis |
RA*, vasculitis, IBD*, RPC* |
| Scleritis |
RA*, GPA*, SpA*, Behcet’s disease, IBD*, PMR*, |
| Scleritis associated with ulcerative keratitis |
RA*, GPA*, RPC*, SLE*, Sjogren’s, Behcet’s disease, PsA, sarcoidosis |
| Necrotizing scleritis without inflammation (scleromalacia perforans) |
Almost exclusively in RA |
| Uveitis |
Behcet’s disease & sarcoidosis can present in any form of uveitis |
| Anterior |
RA/JIA, SpA, HLA-B27* non-specific arthropathy, reactive arthritis, PsA*, GPA*, IBD*, JIA*, Kawasaki disease |
| Intermediate |
Multiple sclerosis |
| Posterior |
PsA*, IBD*, SLE*, GCA*, PAN*, GPA* |
| Panuveitis |
SLE*, VKH*, HLA-B27* associated, RPC*, PAN*, dermatomyositis |
| Retinal vasculitis |
Most commonly Behcet’s disease, sarcoidosis, multiple sclerosis
Less frequently: ANCA associated vasculitis, large and medium vessel
vasculitis, APS*
Other: HLA-B27* associated uveitis dermatomyositis, Takayasu’s,
polymyositis, RPC*, RA* |
| Optic neuropathy |
GCA*, SLE*, APS* |
*GPA: Granulomatosis with polyangiitis; IBD: Inflammatory bowel disease;
RA: Rheumatoid arthritis; SLE: Systemic lupus erythematosus; RPC: Relapsing
polychondritis; SpA: spondyloarthritis; PMR: Polymyalgia rheumatica; JIA: Juvenile idiopathic arthritis;
PsA: Psoriatic arthritis; PAN: polyarteritis nodosa;
VKH: Vogt-Koyanagi-Harada; APS: Antiphospholipid syndrome |
-
Approximately 40% of patients with SpA experience ≥1 EMSK manifestation during the course of the disease: 10,11,12,13
- Acute anterior uveitis (AAU) is one of the common EMSK manifestations of the SpA spectrum of diseases.
- The prevalence of AAU is 22-40% in AS, with lower presentation in the rest of the entities (psoriatic arthritis or reactive arthritis).
- HLA-B27 is present in 50% of AAU patients.
-
Uveitis, when left untreated, is an important cause of blindness14 after under-corrected refractive errors, cataracts, glaucoma, and diabetic retinopathy.15
- It is estimated to cause 10-15% of blindness in the U.S.
-
Always remind yourself about the possible eye–gut connection.16,17
- The prevalence of IBD in AS is about 5-10%.
- The prevalence of ophthalmic inflammatory disorders in IBD is variable, according to the population studied, ranging from 0.3% to 13.0% among all IBD patients.
-
AAU therapy includes topical cycloplegics (i.e., topical atropine), nonsteroidal anti-inflammatory drugs (NSAIDs), corticosteroids (topical, ocular injections, systemic). In recalcitrant cases, agents such as disease-modifying antirheumatic drugs (DMARDs) (i.e., methotrexate), mycophenolic acid, or adalimumab can be highly effective.
-
The most significant side effects of some of the drugs used to treat rheumatic disease are maculopathy associated with anti-malarial agents, and cataracts and glaucoma associated with corticosteroid use.2
-
When referring a patient to ophthalmology or when getting a referral from an ophthalmologist asking to rule out a rheumatologic condition in a patient with an inflammatory eye disease, be mindful of the investigations sent. Not every single condition in rheumatology causes eye involvement, such as uveitis.9,18–20
For example, a diagnostic workup of value in a patient with unclassified uveitis includes routine complete blood count (CBC), comprehensive metabolic panel, urinalysis (UA), erythrocyte sedimentation rate (ESR), and C-reactive protein (CRP). Based on these results and the medical history, infectious disease studies, imaging and autoantibodies may be ordered.
- All patients should have a chest X-Ray, venereal disease research laboratory test (VDRL) and the fluorescent treponemal antibody absorption (FTA-ABS).
- Purified protein derivative (PPD) test and Quanti-FERON: if there is suspicion of exposure to tuberculosis, CXR findings or pre-immunomodulatory therapy (IMT) assessment.
- ANA: only in pediatric patients with pauciarticular JIA and uveitis (prognosis). In adults, only consider if there are other features of SLE, anti-C1q disease or another ANA-associated disease.
- HLA-B27 is appropriate for patients with AAU, even in the absence of demonstrable SpA (prognostic implications).
-
With multiple crossover patients among these two subspecialities,
rheumatologists should not ignore ocular
symptoms and ophthalmologists should be specific in
their referral regarding the ocular diagnosis and the
possible systemic diseases they are suspecting in their
patients. Ideally the two specialities should hold a clinic
together to follow-up and manage these complex patients
together. If a combined clinic is not possible, there
should be a communication tool that allows for clear
communication of patients’ progress and management
between the two of them.
References:
1. Rosenbaum JT, Asquith M. The microbiome and HLA-B27-associated acute anterior uveitis. Nat Rev Rheumatol. 2018; 14(12):704-713. doi:10.1038/s41584-018-0097-2.
2. Hamideh F, Prete PE. Ophthalmologic manifestations of rheumatic diseases. Seminars in Arthritis and Rheumatism. 2001; 30(4):217-241. doi:10.1053/sarh.2001.16639.
3. Petris CK, Almony A. Ophthalmic manifestations of rheumatologic disease: diagnosis and management. Missouri medicine. 2012;109(1):53-58. Accessed May 5, 2021. /pmc/articles/PMC6181687/.
4. Su Y, Yang C. Keratoconjunctivitis Sicca in Sjögren’s Syndrome. New Engl M Med. 2020;383(17):1663-1663. doi:10.1056/nejmicm1910311.
5. Rademacher J, et al. Therapeutic Advances in Musculoskeletal Disease Special Collection. Ther Adv Musculoskelet Dis. 2020 Sep 12;12 doi:10.1177/1759720X20951733.
6. Jayson MI, Jones DEP. Scleritis and rheumatoid arthritis. Ann rheum Dis.1971; 30:343. doi:10.1136/ard.30.4.343
7. Keratoconjunctivitis Sicca: an overview (ScienceDirect Topics). Accessed May 5, 2021.
www.sciencedirect.com/topics/medicine-and-dentistry/keratoconjunctivitis-sicca.
8. Granulomatosis with polyangiitis and microscopic polyangiitis: Clinical manifestations and diagnosis – UpToDate. Accessed May 5, 2021. https://www.uptodate.com/contents/granulomatosis-with-polyangiitis-and-microscopic-polyangiitis-clinical-manifestations-and-diagnosis.
9. Autoimmune Eye & Ear Disorders | Rheumatology Secrets. Accessed May 5, 2021. https://expertconsult.inkling.com/read/west-rheumatology-secrets-4e/chapter-75/autoimmune-eye-and-ear-disorders.
10. van der Heijde D, Ramiro S, et al. 2016 update of the ASAS-EULAR management recommendations for axial spondyloarthritis. Ann Rheum Dis. 2017 Jun; 76(6):978-991. doi:10.1136/annrheumdis-2016-210770.
11. Sieper J, van der Heijde D. Review: Nonradiographic axial spondyloarthritis: New definition of an old disease? Arthritis Rheum. 2013; 65(3):543-551. doi:10.1002/art.37803
12. Wakefield D, Clarke D, McCluskey P. Recent Developments in HLA B27 Anterior Uveitis. Frontiers in Immunology. 2021; 11:3380. doi:10.3389/fimmu.2020.608134.
13.Baarsma GS. The epidemiology and genetics of endogenous uveitis: A review. Current Eye Research. 1992; 11(S1):1-9. doi:10.3109/02713689208999505.
14. The Ocular Immunology and Uveitis Foundation. uveitis.org. Accessed May 5, 2021. https://uveitis.org/patients/education/glossary/g-l/.
15. Bourne RRA, Steinmetz JD, Saylan M, et al. Causes of blindness and vision impairment in 2020 and trends over 30 years, and prevalence of avoidable blindness in relation to VISION 2020: The Right to Sight: An analysis for the Global Burden of Disease Study. The Lancet Global Health. 2021; 9(2):e144-e160. doi:10.1016/S2214-109X(20)30489-7.
16. Troncoso LL, Biancardi AL, de Moraes HV, et al. Ophthalmic manifestations in patients with inflammatory bowel disease: A review. World J of Gastroenterol. 2017; 23(32):5836-5848. doi:10.3748/wjg.v23.i32.5836.
17. Stolwijk C, van Tubergen A, Castillo-Ortiz JD, et al. Prevalence of extra-articular manifestations in patients with ankylosing spondylitis: A systematic review and meta-analysis. Ann Rheum Dis. 2015; 74(1):65-73. doi:10.1136/annrheumdis-2013-203582.
18. Bouzid N, Jamilloux Y, Chapurlat R, et al. Impact of systemic treatments on the course of HLA-B27-associated uveitis: A retrospective study of 101 patients. PLoS ONE. 2020; 15(3). doi:10.1371/journal.pone.0230560.
19. Brewerton DA, Hart FD, Nicholls A, et al. Ankylosing Spondylitis and HLA-27. The Lancet. 1973; 301(7809):904-907. doi:10.1016/S0140-6736(73)91360-3.
20. Park SC, Ham D. Clinical features and prognosis of HLA-B27 positive and negative anterior uveitis in a Korean population. J Korean Med Sci. 2009; 24(4):722-728. doi:10.3346/jkms.2009.24.4.722
Vanessa Ocampo, MD, FRCPC
Psoriatic arthritis fellow, Center for Prognosis of Rheumatic Diseases
University of Toronto, Toronto, Ontario
|




