Spring 2021 (Volume 31, Number 1)
COVID-19:
Innovations in Delivering
Science to Rheumatologists
and Patients
By Sindhu R. Johnson, MD, PhD
Download PDF

In order to efficiently
meet the needs of our
patients during the
COVID-19 pandemic, innovations
in the way we
conduct science and disseminate
new knowledge
have emerged. In my capacity
as Chair of the American
College of Rheumatology
(ACR) Quality of Care
Committee, I have had the
opportunity to participate
in or oversee a few COVID-19 initiatives.
Global Rheumatology Alliance
The Global Rheumatology Alliance (GRA) is a grassroots
organization with origins in social media and a vision of
“bringing together the global rheumatology community
to curate and disseminate accurate and comprehensive
knowledge to advance rheumatology care in the
COVID-19
pandemic.” Many Canadian rheumatologists have been
involved in this effort, in conjunction with the Canadian
Rheumatology Association (CRA), including Dr. Diane Lacaille,
Dr. Marie Hudson, Dr. Carter Thorne, Dr. Evelyn
Sutton and Dr. Louis Bessette. To achieve this vision, the
GRA has four research arms:
- Provider registries
- Patient experience survey
- Systematic reviews
- Patient telemedicine survey
In a year, this collaboration has been shockingly productive.
The ability to leverage social media to bring people together,
to collect data, to write collaboratively, and publish needed information
in a short time period is remarkable. The GRA is a
new model of not only doing business, but of doing science.
For a list of COVID-19 related publications from the GRA and
ACR, visit rheum-covid.org/publications/ and rheumatology.org/Practice-Quality/Clinical-Support/COVID-19-Guidance.
ACR COVID-19 Vaccine Clinical Guidance Task Force
In 2019, the ACR Board approved the creation of the Guidance
Subcommittee, charged with overseeing the development
of guidance documents for clinical areas the membership
wanted guidance on; areas for which the evidence is insufficient
or timeline too short for more formal, GRADE-process
driven guidelines. The ACR Guidance subcommittee includes
Canadian rheumatologists Dr. Shahin Jamal and Dr. Alex Legge.
The ACR convened two task forces to address the needs
of rheumatology providers during the COVID‐19 pandemic,
in which I had leadership roles. First, the COVID-19
Clinical Task Force was charged by ACR leadership to rapidly
provide guidance to rheumatology providers relevant
to the management of rheumatic disease in adult patients
during the pandemic. Clinical guidance generated from
this effort was intended to aid in the care of individual patients,
but not meant to supplant clinical decision‐making.
Early in the pandemic, this document provided guidance
on the use of rheumatic disease treatments including ACE
inhibitors, ARBs, NSAIDs, glucocorticoids, and immunosuppressives
following known SARS–CoV‐2 exposure and
in the context of active or presumptive COVID-19.
Second, the ACR COVID-19 Vaccine Clinical Guidance
Task Force was struck, consisting of North American rheumatologists,
infectious disease specialists and public health
experts with current or past employment at the Center for
Disease Control and Prevention (CDC). Using a balance of
consensus-based methods and largely indirect evidence from
the literature, guidance on the use of the COVID-19 vaccine
in individuals with rheumatic and musculoskeletal diseases
(RMD), and in particular individuals with autoimmune and
inflammatory rheumatic disease (AIIRD) was created. Topics
included risk of COVID-19 infection and outcomes in RMD
patients, vaccine immunogenicity in the setting of active disease
or immunosuppressive therapy and vaccine safety. No
evidence was found to support a concern regarding the use or
timing of immunomodulatory therapies in relation to mRNA
vaccine safety. Therefore, guidance regarding immunomodulatory
medication and vaccination timing was given considering
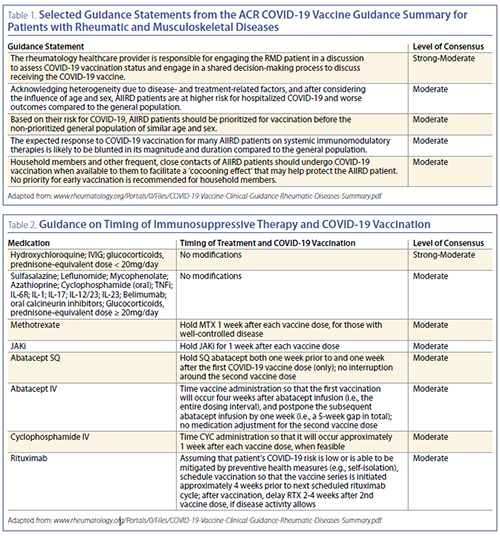
the intent to optimize vaccine response. Highlights from
the ACR COVID-19 Vaccine Clinical Guidance are summarized
in Tables 1 and 2. The draft summary was approved by
the ACR Board of Directors on February 8, 2021; and a full
manuscript is pending journal peer review.
Dissemination of rapidly changing information
Given that information regarding the intersection of RMD,
risk of
COVID-19 infection and outcomes, and immunogenicity/
safety of the COVID-19 vaccine is rapidly emerging, mechanisms to update and release new information needed
to be developed. In his capacity as Associate Editor
at the journal Arthritis and Rheumatology, Dr. Brian Feldman
has been integral to the rapid review and dissemination
of COVID-related manuscripts from the ACR Quality of
Care Committee. Using a new model, the ACR COVID-19
Guidance documents are considered “living documents.”
As new information is published, the guidance documents
are updated.
In short, Canadian rheumatologists have been active
participants in a variety of facets related to the conduct
and dissemination of science during the COVID-19 pandemic.
We will continue to work together for the betterment
of the patients we serve.
Sindhu R. Johnson, MD, PhD
Associate Professor of Medicine, University of Toronto
Clinician-Scientist, Toronto Western Hospital, Mount Sinai Hospital
Associate Director,
Clinical Epidemiology & Health Care Research Program,
Institute of Health Policy, Management and Evaluation
Toronto, Ontario
|




