Winter 2014 (Volume 24, Number 4)
Current Gout Management Practices By Primary-care Physicians in Canada
By Gregory Choy, MD, FRCP;
Niloofer Baria, MD, CCFP;
Alan Bell, MD, CCFP;
Lydia Hatcher, MD, CCFP, FCFP, CHE, CAPM;
Denise Sequeira, MD;
and Daniel Gagnon, MD
Download PDF
Background
Gout affects nearly 3% of adults in Canada.1,2 Several studies have revealed that patients with serum uric acid (SUA) levels lower than
360 µmol/L showed improved outcomes, including reductions in the frequency of attacks,3,4 reductions in tophus area,5,6 and depletion of urate crystal stores in synovial fluid.5,7,8 Prophylactic anti-inflammatory agents should be prescribed with the initiation of chronic urate-lowering therapy (ULT) to avoid or to reduce the risk of mobilization flares.6 Inadequate control of hyperuricemia in gout may have a significant economic impact in society due to both direct medical costs and indirect costs.9-11
Gout is managed mostly by primary care physicians.12,13 The European League Against Rheumatism (EULAR) had developed evidence-based recommendations for the management of gout prior to our study being undertaken.6,14-17 As the new American College of Rheumatology (ACR) 2012 guidelines were published after this study was completed, they were not taken into consideration in this study.
This chart audit program was designed to monitor how gout has recently been managed within Canadian primary-care settings and to identify any care gaps.
Method
This program consisted of a Canada-wide retrospective multicentre observational clinical chart review on gout. Between September 2011 and January 2012, 500 outpatients with gout attending primary care clinics were profiled. General practitioners/family physicians (n = 72) were each asked to complete 10 case report forms based on the first 10 consecutive patients who visited their office, had documented gout, and satisfied the selection criteria requirements. Inclusion criteria included patients above 18 years of age (both male and female) with a history of gout for at least one year. Patients undergoing cancer therapy were excluded. Each physician completed a structured case-report form questionnaire online. The report form questionnaire included patient demographics, burden of illness (SUA levels, number of flares, and symptoms), comorbidities, and treatment. Participating physicians were initially asked to recall the target SUA level prior to initiating ULT treatment. All patients profiled were asked to provide their informed patient consent. This study has been reviewed and approved by Institutional Review Board Services (IRB Services). Current gout treatment practices were benchmarked with EULAR guidelines for the management of gout. Data was collected using a standardized data abstraction form, and was analysed and compared using confidence intervals (CI) and Student’s unpaired 2-tailed t-test.
Participant Demographics and Patient Baseline Characteristics
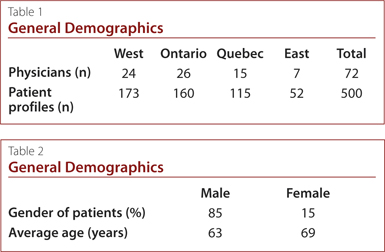
Overall, 72 family physicians were recruited across Canada and
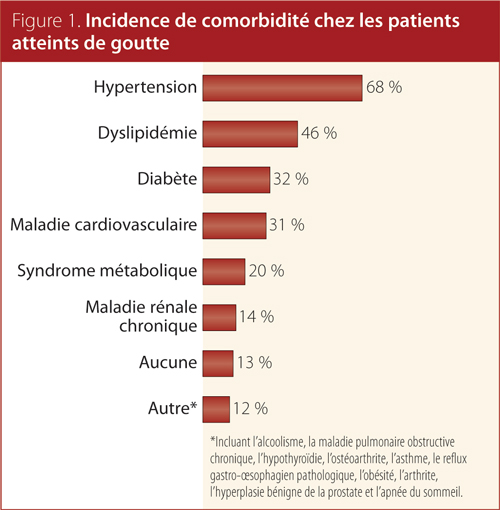
500 patient profiles were completed. The main socio-demographic and medical characteristics of the study cohort are presented in Tables 1 and 2, and Figure 1. Approximately 85% of patients were male and the average age was 64 years old, with a male-to-female ratio of 6:1. The proportion of patients diagnosed with chronic gout was nearly the same across Canada (58%). On average, patients had their SUA levels measured twice a year since initiation of their current therapy and experienced one flare in the previous 12 months. Approximately 75% of chronic gout patients in this cohort received ULT. The vast majority of patients (61%) were receiving allopurinol, with 11% receiving febuxostat. Nearly 60% of gout patients were taking anti-inflammatory medications over an average period of eight weeks.
Standard Target SUA Levels
Approximately two-thirds of the physicians surveyed across Canada claimed to have a standard therapeutic SUA target. However, only 38% of the respondents were treating gout patients to an optimal SUA target (SUA level < 360 µmol/L). Nearly 45% of the physicians were treating gout patients suboptimally or were not aware of a therapeutic target.
Physicians treating to an optimal target tend to have an increased number of patients with fewer than three flares in the last year (43%), compared to those (56%) who are not treating to target. The average SUA measurement was significantly lower in patients who had experienced fewer than three flares in the previous 12 months (378 µmol/L compared to 467 µmol/L, p ≤ 0.004). Overall, these results showed that patients with non-target SUA levels were 56% more likely to flare than those at target.
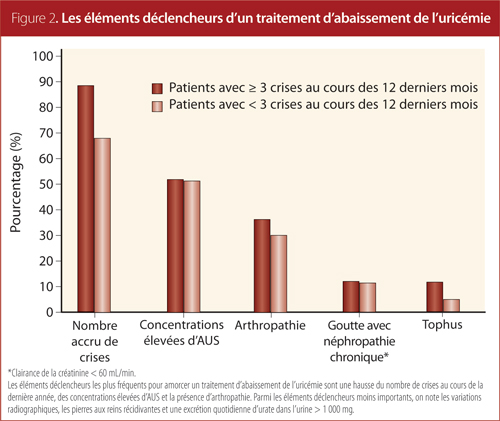
Triggers for Initiating ULT
In the current study, several factors motivated family physicians to initiate ULT therapy for gout patients (Figure 2), with increasing number of flares over past year being the most common.
Duration of Anti-inflammatory Prophylaxis in Conjunction With ULT
Patients treated to an optimal target were more likely to be prescribed anti-inflammatory prophylaxis (64%) relative to patients suboptimally treated (54%). Physicians prescribed anti-inflammatory prophylaxis to patients on ULT for an average period of eight weeks, which is below the guideline duration (six months) recommended by the EULAR task force.
Discussion
To our knowledge, this chart audit program is the first study conducted in Canada undertaken to examine practice patterns in the management of gout, as well as the quality of care received by gout patients from family doctors.
This Canadian study population showed demographics similar to those in the US and UK, with men in their 60s being the most common group. Over 70% were on ULT; allopurinol remained the most common agent and febuxostat accounted for approximately 10% of usage.
A significant proportion of primary-care physicians were not treating gout to target therapeutic SUA levels. The patients who were not treated to target experienced more flares overall. In addition, patients were often not prescribed anti-inflammatory prophylaxis, or the duration of time on prophylaxis was too short. This would also lead to more post-initiation flares.
This study had several limitations. Physicians were invited randomly to participate in the program. Those who participated might have a higher level of interest in gout management, which could have influenced our results as they may not be representative of the common primary care physician population. A small sample size (n = 500) and the retrospective nature of chart audits also limited statistical analysis.
Overall, this study highlights the need to educate physicians on the importance of treating gout more aggressively. With no recently updated Canadian guidelines in gout management, it was unclear which set of evidence-based guidelines had been followed by primary care physicians in Canada. The etiology of gout is well characterized, and effective therapies are available for both acute and long term management. Better dissemination of best practice guidelines would be important in
improving quality of treatment for patients with gout.
References
1. Wong R, Davis AM, Badley E, et al. Prevalence of Arthritis and Rheumatic Diseases Around the World. A Growing Burden and Implications for Health Care Needs.2010. MOCA2010-07/002.
2. Badley E, DesMeules M. Health Canada. Arthritis in Canada. An Ongoing Challenge. Ottawa.2003. H39-4/14-2003E.
3. Zhang W, Doherty M, Pascual E, et al. EULAR evidence based recommendations for gout. Part I: Diagnosis. Report of a task force of the Standing Committee for International Clinical Studies Including Therapeutics (ESCISIT). Ann Rheum Dis 2006; 65(10):1301-11.
4. Perez-Ruiz F. Treating to target: a strategy to cure gout. Rheumatology (Oxford) 2009; 48 Suppl 2:ii9-ii14.
5. Becker MA, Schumacher HR Jr., Wortmann RL, et al. Febuxostat compared with allopurinol in patients with hyperuricemia and gout. N Engl J Med 2005; 353(23):2450-61.
6. Shoji A, Yamanaka H, Kamatani N. A retrospective study of the relationship between serum urate level and recurrent attacks of gouty arthritis: evidence for reduction of recurrent gouty arthritis with antihyperuricemic therapy. Arthritis Rheum 2004; 51(3):321-5.
7. Doherty M. New insights into the epidemiology of gout. Rheumatology (Oxford) 2009; 48 Suppl 2:ii2-ii8.
8. Pascual E, Sivera F. Time required for disappearance of urate crystals from synovial fluid after successful hypouricaemic treatment relates to the duration of gout. Ann Rheum Dis 2007; 66(8):1056-8.
9. Pascual E, Sivera F. Why is gout so poorly managed? Ann Rheum Dis 2007; 66(10):1269-70.
10. Doherty M, Jansen TL, Nuki G, et al. Gout: why is this curable disease so seldom cured? Ann Rheum Dis 2012; 71(11):1765-70.
11. Jordan KM, Cameron JS, Snaith M, et al. British Society for Rheumatology and British Health Professionals in Rheumatology guideline for the management of gout. Rheumatology (Oxford) 2007; 46(8):1372-4.
12. Zhang W, Doherty M, Bardin T, et al. EULAR evidence based recommendations for gout. Part II: Management. Report of a task force of the EULAR Standing Committee for International Clinical Studies Including Therapeutics (ESCISIT). Ann Rheum Dis 2006; 65(10):1312-24.
13. Mikuls TR, Farrar JT, Bilker WB, et al. Suboptimal physician adherence to quality indicators for the management of gout and asymptomatic hyperuricaemia: results from the UK General Practice Research Database (GPRD). Rheumatology (Oxford) 2005; 44(8):1038-42.
14. Roddy E, Zhang W, Doherty M. The changing epidemiology of gout. Nat Clin Pract Rheumatol 2007; 3(8):443-9.
15. Perez-Ruiz F, Calabozo M, Pijoan JI, et al. Effect of urate-lowering therapy on the velocity of size reduction of tophi in chronic gout. Arthritis Rheum 2002; 47(4):356-60.
16. Li-Yu J, Clayburne G, Sieck M, et al. Treatment of chronic gout. Can we determine when urate stores are depleted enough to prevent attacks of gout? J Rheumatol 2001; 28(3):577-80.
17. Cronstein BN, Terkeltaub R. The inflammatory process of gout and its treatment. Arthritis Res Ther 2006; 8 Suppl 1:S3.
Gregory Choy, MD, FRCP
Interim Head,
Division of Rheumatology,
Sunnybrook Health Sciences Centre
Toronto, Ontario
Niloofer Baria, MD, CCFP
Director of Foundations of Medicine,
MD Undergraduate Education
University of British Columbia
Family Physician
Vancouver, British Columbia
Alan Bell, MD, CCFP
Assistant Professor,
Department of Family and Community Medicine,
University of Toronto
Toronto, Ontario
Lydia Hatcher, MD, CCFP, FCFP, CHE, CAPM
Family Physician,
Family Wellness Place
Mount Pearl, Newfoundland
Denise Sequeira, MD
Medical Science Liaison,
Takeda Canada Inc.
Oakville, Ontario
Daniel Gagnon, MD
Rheumatologist,
Centre Médical et Chirurgical Rockland
Montreal, Quebec
|



