Spring 2014 (Volume 24, Number 1)
A Call To Action For Adult Vaccination: Immunocompromised Patients At Increased Risk
By Carolyn Whiskin, RPh, BScPhm, NCMP;
Derek Haaland, MD, MSc, FRCPC;
William Bensen, MD, FRCPC; and
Vivien Brown, MDCM, CCFP, FCFP, NCMP
Download PDF
The Issue
Pediatric vaccination in Canada experiences high levels of adherence due to well-established protocols, public health support, and school-based programs. The same cannot be said for adult immunization. There is no established “check point” for adults regarding updating their vaccines and, according to the 2006 Canadian Immunization Survey, most adult Canadians are under-immunized for all vaccines. It has been suggested that a good time for this preventative health care discussion may occur at age 50 when adult screening tests for bowel and breast cancers, amongst others, begin.
This is not yet the current standard practice in Canada; as such, when patients require treatments which may suppress their immune system, specialists prescribing these therapies cannot assume that their patient’s vaccinations have been updated. At the time of treatment initiation, the question becomes: “Who is responsible for ensuring vaccinations are given?” While the family physician has traditionally been responsible for advising on the use and administration of vaccines, specialists initiating treatment that will suppress the immune system have a level of responsibility to discuss and ensure their patients are vaccinated before treatment initiation. At the CRA Annual Scientific Meeting (ASM) in 2013, rheumatologists were surveyed regarding vaccinations. Results indicated that, although a discussion regarding vaccination was considered important by the majority of those surveyed, it was not part of regular practice. Reasons for not discussing vaccination included the amount of other necessary information to be discussed regarding diagnosis and treatment options, the patient being overwhelmed with information, and lack of time and nursing support. While it is important to note that adult vaccination may not have the same impact in preventing disease as it does in children, it has significant impact on attenuating disease in adults, decreasing morbidity and mortality rates. Our current statistics are dramatic as the death rate from vaccine preventable disease is 100-fold increased in adults compared to children.
What is the Current Status In An At-risk Population?
A retrospective population-based cohort of rheumatoid arthritis (RA) patients aged 66 years and older was assembled using Ontario health administrative data from April 1, 1992 to March 1, 2010. All patients were required to have had at least one exposure to a disease-modifying antirheumatic drug (DMARD), biologic agent, or oral glucocorticoid at the time of cohort entry. Infection rates were found to be higher in this population compared to the general population, with the highest incidence of infection seen for bacterial pneumonia, herpes zoster, and skin or soft tissue infections. When vaccinations are not given prior to administration of immunosuppressant agents, the already vulnerable patient is left unprotected. The consequence of acquiring pneumonia or shingles is beyond the burden of the infection itself, as patients are required to withhold their primary treatment until the infection clears. This has the potential to cause a relapse of the condition being treated; in the case of biologic treatment, missed doses can increase immunogenicity and result in a decrease in treatment effectiveness and increased adverse effects. Patients described loss of remission and increased disease burden of the initial chronic disease long after the secondary illness resolved. In a biologic clinic in Hamilton, Ontario, six cases of shingles were diagnosed within a one-month period, all resulting in withholding treatment. The resultant post-herpetic neuralgia along with arthritic flares were impactful for these patients. Not one of the six cases had received a zoster vaccination and none reported their physician discussing vaccination before treatment initiation.
Invasive pneumococcal disease (IPD) due to streptococcus pneumonia has the highest incidence rate in the young and those over 50 years of age; risk of death due to IPD also increases with age. The incidence of shingles within a patient’s lifetime is 30%, which also increases with age. Of greatest concern is the percentage of patients with post-herpetic neuralgia lasting greater than one year; rates have been shown to rise from 15% to 40% going from age 50 to older than age 70. The question is not only “What is the cost of vaccination?”, but also, “What is the cost of not being vaccinated?”
A Call To Action
The CRA has published guidelines regarding appropriate use of vaccines prior to and during DMARD/biologic therapy. In 2013 the National Advisory Committee on Immunization (NACI) guidelines were updated with regards to immunocompromised patients. However, guidelines must be implemented to be effective. At The Charlton Centre for Specialized Treatment in Hamilton, Ontario, a protocol has been developed based on these guidelines. The protocol was launched in April 2013 by protocol authors Carolyn Whiskin and Dr. Jay Keystone. Since that time, referring physicians to the clinic have all received paper and electronic versions of the protocol (see Appendix 1). The Clinic’s director of pharmacy programs, Carolyn Whiskin, subsequently visited each referring physician to discuss implementation of the protocol into their practice. Some rheumatologists have incorporated the protocol recommendations into their referral note to the Primary Health Care Provider along with providing a copy for the patient to share on their next visit to their family doctor. Other specialists have included vaccine discussion as part of a checklist for new patients entering their practice and built it into their electronic medical records. Some rheumatologists have taken the initiative to prescribe the vaccines not covered under government funding (e.g., the conjugated pneumococcal vaccine [PPV13] and the herpes zoster vaccine), as these are both highly recommended for patients who will be receiving biologic DMARDS. Ideally, the discussion regarding vaccination needs to happen at the time of diagnosis, rather than waiting until biologic treatment is prescribed. At the Charlton Clinic, patients are screened when a prescription for biologic treatment is received. If this discussion has been missed, a window of opportunity exists during the time that reimbursement is being coordinated for vaccinations to be updated. A cover letter to accompany the vaccine protocol can be sent to the patient’s family physician at the request of the specialist prescribing immunosuppressant therapy and the patient.
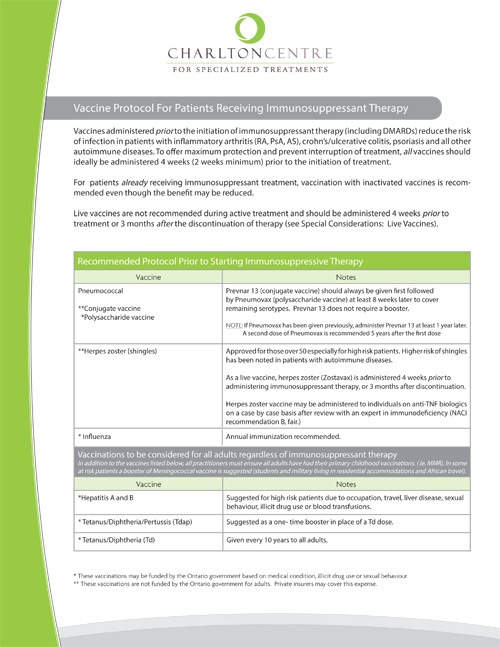

While uptake of the protocol at some rheumatologists’ offices has been slow to be implemented, others have gone from no vaccination discussion to 100% of patients receiving information and following up with their family physician. The herpes zoster vaccine is not a covered benefit in any province; PPV13, although a benefit for children, has limited coverage across Canada for at-risk patients. It is important to note that provincial regulations are under regular revision. As there is a cost associated with some vaccines, the decision to pursue vaccination is still patient based, with only 30% of private insurers covering the expense.
Infectious disease prevention through the vaccination of immunosuppressed patients makes sense biologically, given that any relative risk reduction will have the biggest pay-off in a group with high baseline risk. That said, infection risk and vaccine responses in a given individual are multifactorial, with both disease-related and medication-related factors playing a role. In fact, in terms of infection risk, there is evidence that active disease may play a bigger role in immunocompromised patients than disease-modifying treatment itself. Thus, rheumatoid disease control via immunosuppression, somewhat paradoxically, may well be the single most important factor in infection reduction.
Coordinated multicentre efforts will be required to delineate the clinical effects of vaccination in our immunosuppressed population. Studies on immunogenicity of vaccines in such patients will be relevant, but even more important will be the long-term clinical follow up of large numbers of patients to track infection incidence.
The time has come to take action. The role of rheumatologists is expanding and the use of immunosuppressive medication requires not only a fully informed patient, but also a fully vaccinated patient to reduce inherent, preventable risk. As we have national guidelines from NACI and the CRA, it is becoming increasingly clear that the standard of care in Canada includes aggressive prevention of infections in immunosuppressed patients. Our role is to advocate, facilitate, and ultimately vaccinate our population if we are to achieve the outcomes of disease modification that we seek in our at-risk, adult population.
Suggested Readings
1. Ruibin LG, Levin MJ, Ljungman P, et al. 2013 IDSA Clinical Practice Guideline for Vaccination of the Immunocompromised Host. Clin Infect Dis 2014; 58(3):309-18.
2. Listing J, Gerhold K, Zink A. The risk of infections associated with rheumatoid arthritis, with its comorbidity and treatment. Rheumatology (Oxford) 2013; 52(1):53-61.
3. Public Health Agency of Canada. An Advisory Committee Statement (ACS) - National Advisory Committee on Immunization (NACI): Update on the Use of Herpes Zoster Vaccine. 2014. Available from: www.publications.gc.ca/collections/collection_2014/aspc-phac/HP40-92-2014-eng.pdf
Carolyn Whiskin, RPh, BScPhm, NCMP
Pharmacy Program Director,
The Charlton Centre For Specialized Treatment
Hamilton, Ontario
Derek Haaland, MD, MSc, FRCPC
Assistant Clinical Professor,
McMaster University
Divisions of Clinical Immunology & Allergy and Rheumatology
Shanty Bay, Ontario
William G. Bensen, MD, FRCPC
Clinical Professor,
Division of Rheumatology,
Department of Medicine,
McMaster University
Hamilton, Ontario
Vivien Brown, MDCM, CCFP, FCFP, NCMP
Assistant Professor,
Department of Family and Community Medicine,
University of Toronto
Vice President,
Medical Affairs,
Medisys Corporate Health
Toronto, Ontario |



